A technical decision framework for engineers weighing boron nitride against graphite when graphite keeps failing in service.
Graphite has been the default melting vessel for decades — affordable, machinable, stable under inert cover. Then a process variable shifts. Air exposure creeps above 500°C. The melt starts accepting carbon. Downstream GDMS flags contamination that traces back to the crucible wall. At that point the engineer proposes boron nitride, and procurement pushes back on the 3-5× price gap.
The decision is rarely about which material performs better. Almost every datasheet tells that story. The real question sits on three axes: atmosphere, melt chemistry, and purity tolerance. When any one of them crosses a threshold, graphite stops being an option regardless of cost. When all three stay inside graphite's envelope, BN's premium never recovers.
This article draws the thresholds, quantifies the payback where it exists, and closes with an RFQ specification engineers can hand to procurement this week.
Boron nitride crucibles justify a 3-5× price premium over graphite when operating in oxidizing atmospheres above 500°C, processing carbon-sensitive melts such as gallium, aluminum at superheat, or III-V compounds, or targeting sub-10-ppm metallic purity. For inert-atmosphere melting of carbon-tolerant metals — copper, brass, gold, silver — graphite remains the rational choice and the BN premium does not recover. The decision is binary on chemistry and atmosphere; budget enters only after technical necessity is confirmed.
BN justifies the premium in three operating windows:
- Oxidizing atmospheres above 500°C — graphite oxidizes steadily; BN remains stable to ~850°C in air per JIS R1648.
- Carbon-sensitive melts — graphite contributes ppm-level carbon to gallium, aluminum at superheat, alkali metals, titanium aluminides, and III-V compounds; BN contributes none.
- Sub-10-ppm metallic purity targets — MBE source material, single-crystal growth, and photovoltaic precursor refining cannot tolerate graphite carbon flux at any temperature.
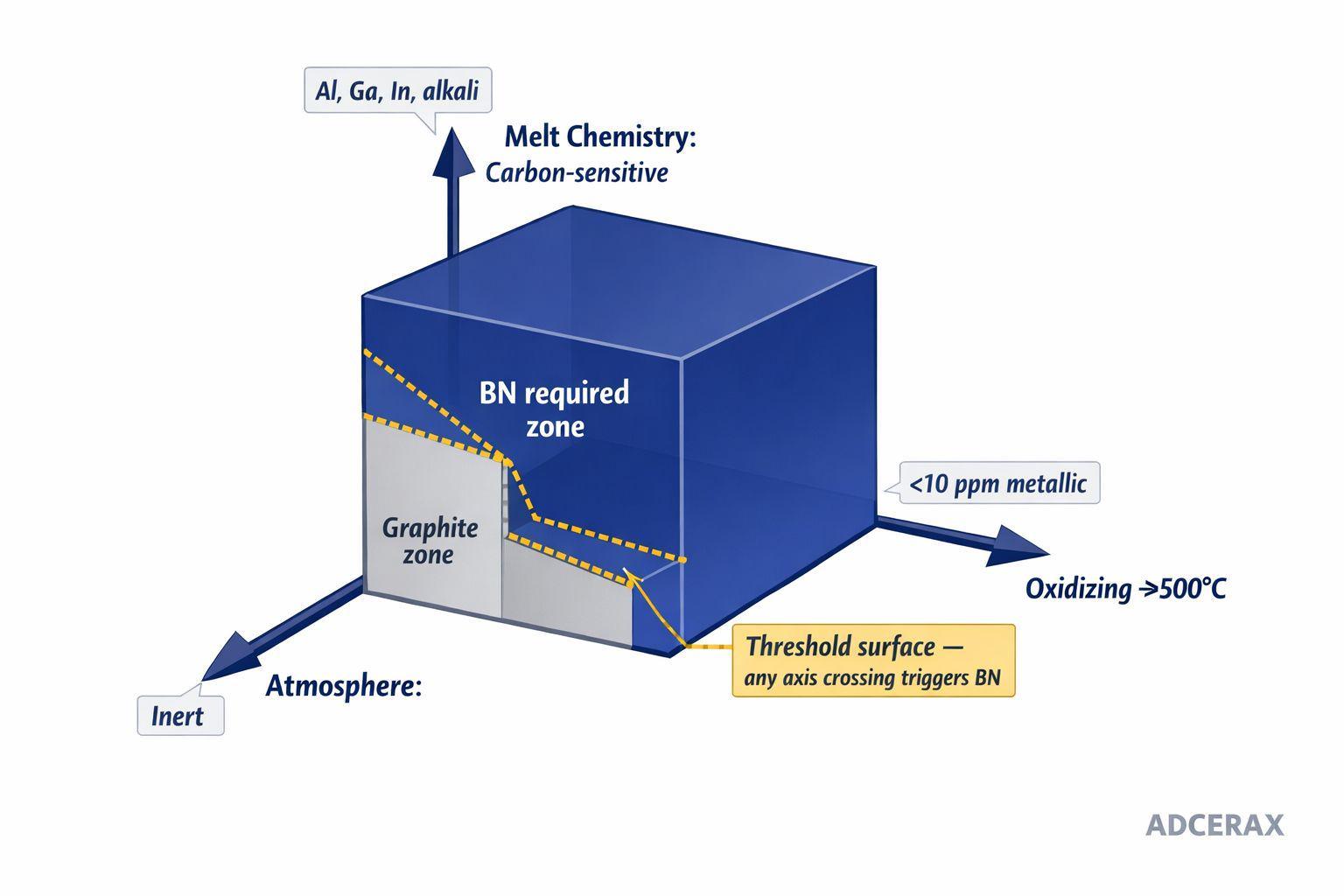
The BN-required zone is defined by three crossing thresholds — reading the operating point on this map is faster than any cost argument.
Why does my graphite crucible keep failing — and would BN actually solve it?
Graphite crucible failure in industrial service follows four mechanisms: atmospheric oxidation above ~500°C, carbon dissolution into reactive melts, metal wetting and wall infiltration, and thermal-mechanical fatigue from repeated cycling. Boron nitride eliminates the first three categorically — it resists air oxidation to ~850°C per JIS R1648, contributes no carbon, and exhibits genuine non-wetting behavior with most molten metals. It does not solve impact damage, improper handling, or operations where graphite was already succeeding. The failure category has to be identified before BN is treated as the answer.
At-a-glance property comparison before the failure-mechanism analysis below:
| Property | Graphite | Boron Nitride (HBN) |
|---|---|---|
| Max operating T in air | ~500°C (oxidation begins) | ~850°C (per JIS R1648) |
| Max operating T in inert / vacuum | ~2,800°C (short hold, inert) | ~1,800°C inert / ~2,100°C vacuum |
| Carbon contribution to melt | ppm to percent-level dissolution | None (carbon-free) |
| Wetting by molten metals | Wets Al at superheat, Ga, In, alkali metals | Non-wetting with most molten metals and slags |
| Typical cycles (carbon-sensitive service) | 50–150 | 300–800 |
| Bulk density range | 1.7–1.9 g/cm³ | 1.9–2.1 g/cm³ |
| Acquisition cost (relative index) | 1.0× | 3.0–5.0× |
Temperature data cross-referenced with JIS R1648 oxidation behavior; cycle-life ranges are field-observed for carbon-sensitive melting service.
Four failure modes of graphite crucibles in industrial service
Oxidation is the most visible. Above roughly 500°C in air, graphite loses mass steadily; wall thickness thins, and service life drops from hundreds of cycles to tens. Protective coatings or a strict inert cover are the only mitigations.
Carbon dissolution is the most expensive. Molten aluminum at superheat, gallium, indium, alkali metals, titanium aluminides, and several rare-earth alloys pick up carbon from the crucible wall at ppm-to-percent levels. The contamination moves downstream, often reaching final product before quality flags it.
Wetting and infiltration cause molten metal to climb the crucible wall and seep through interconnected porosity. On cooldown the charge locks into the vessel, cracking the crucible and sometimes damaging the furnace lining.
Thermal-mechanical fatigue closes the list. Graphite grain orientation accommodates strain poorly under aggressive ramp rates, and micro-cracks propagate cycle by cycle until wall failure.
Which failures BN eliminates — and which it does not
Boron nitride is chemically inert to carbon-sensitive melts, resists oxidation in air to ~850°C, and its basal-plane structure produces the non-wetting behavior that graphite lacks. Those three failure modes disappear.
Post-mortem analysis of failed graphite crucibles in aluminum superheat service typically shows aluminum carbide (Al₄C₃) deposits on the inner wall and metal climb extending two-thirds up the wall — both signatures of carbon dissolution and wetting that BN eliminates categorically.
What BN does not solve: drop impact (BN is more brittle than graphite), moisture pickup in unsealed storage, and applications already operating inside graphite's safe envelope. For readers whose failure is not BN-solvable, compare crucible material options across oxide and non-oxide families before committing to a grade change.
Diagnostic checklist: is your failure BN-solvable?
Run three questions against the failed crucible. Was the service atmosphere oxidizing or air-exposed? Did downstream analysis detect carbon contamination? Did post-mortem show metal climbing the wall or infiltrating porosity? One or more ""yes"" answers points to BN. All ""no"" answers points to a different root cause — grade change, process tuning, or handling discipline.

The physical signature of carbon pickup — residue and wall climb on graphite, versus the clean non-wetting interior BN leaves after the same service.
At what temperature, atmosphere, and melt chemistry does BN become non-negotiable?
The operating boundary is a three-variable surface, not a single temperature. Any oxidizing or air-exposed process above ~500°C removes uncoated graphite from continuous service, while boron nitride crucibles maintain structural stability up to approximately 850°C in air and 1,800°C in inert atmospheres. Any melt that dissolves carbon — aluminum at superheat, gallium, indium, alkali metals, Ti-Al intermetallics — makes graphite unusable regardless of temperature. Applications targeting below 10 ppm metallic purity fall outside graphite's envelope even when chemistry would otherwise permit it.
The table below translates the three-axis boundary into go/no-go decision rows:
| Operating Variable | Graphite Acceptable | BN Required | Either Viable |
|---|---|---|---|
| Atmosphere | Inert / vacuum / reducing | Air or oxidizing >500°C | Inert <1,500°C |
| Melt chemistry | Cu, brass, Au, Ag, Zn, Pb | Al (superheat), Ga, In, alkali metals, Ti-Al, III-V compounds | Fe (short cycle, inert) |
| Purity target | >100 ppm metallic OK | <10 ppm metallic, carbon-free required | 10-100 ppm |
| Peak temperature | ≤2,800°C (inert, short hold) | ≤850°C air / ≤1,800°C inert / ≤2,100°C vacuum | 1,000-1,500°C inert |
| Process examples | Foundry casting, jewelry melting | MBE effusion cell, PV silicon, GaAs growth, Al refining | Lab-scale alloy melting |
Temperature ranges cross-referenced with JIS R1648 oxidation behavior and industrial vacuum-melting practice.
Carbon-sensitive melts — the concrete list
Reactive and carbon-soluble melts span more of the periodic table than most engineers assume. The industrially significant set: aluminum melt refining above ~700°C, gallium evaporation for photovoltaic and compound-semiconductor feedstock, indium for III-V compound semiconductor growth, alkali metals across the board, titanium aluminides, and rare-earth alloys feeding single-crystal growth.
Photovoltaic silicon precursor refining and gallium evaporation both fail in graphite for the same reason — measurable carbon transport across the crucible-melt interface. These applications do not tolerate graphite at any price.
Where graphite wins — applications BN should not displace
Inert-atmosphere melting of copper, brass, gold, silver, and zinc sits firmly inside graphite's envelope. Carbon tolerance is high, temperatures stay below oxidation limits under cover gas, and throughput economics favor graphite's lower acquisition cost and easier machining. Foundry casting and jewelry melting rarely benefit from BN.
Vacuum induction furnace work with short cycles on carbon-tolerant metals is another graphite-preferred window — BN's oxidation advantage disappears in vacuum, and its cost does not.
For engineers whose operating point sits inside the BN-required zone, BN crucible specifications are the next document to review before drafting an RFQ.
How does the 3-5x acquisition premium reconcile against total cost of ownership?
At the unit-acquisition level the 3-5× premium is real. At total cost of ownership the relationship inverts in three common scenarios: replacement frequency, contamination rework, and manual cleaning labor. In carbon-sensitive melting service, BN crucibles routinely deliver 300 to 800 thermal cycles versus 50 to 150 for graphite, inverting the per-cycle cost despite the higher acquisition price. In applications where graphite already delivers normal life with no contamination penalty, TCO favors graphite permanently — the BN premium never recovers.
The TCO comparison for a carbon-sensitive melt scenario decomposes as follows:
| Cost Element | Graphite Crucible | BN Crucible | Notes |
|---|---|---|---|
| Acquisition cost (relative index) | 1.0× | 3.0–5.0× | Function of grade and geometry |
| Typical cycles to failure (carbon-sensitive service) | 50–150 | 300–800 | Field-observed range; varies by chemistry |
| Per-cycle vessel cost | High | 30–60% of graphite per-cycle | BN advantage emerges from longevity |
| Contamination rework risk | Recurring | Eliminated | Single off-spec batch typically > BN premium |
| Manual cleaning labor between charges | Required | Minimal (non-wetting) | Significant in small-batch operations |
| TCO outcome | Higher in carbon-sensitive service | Lower in carbon-sensitive service | Inverts when chemistry/atmosphere is benign |
Based on field analysis of industrial crucible installations. Actual ratios vary by process; calculate with site-specific cycle counts.
TCO formula with worked example
Three numbers drive the calculation: graphite cycles-to-failure in your actual service (Cg), BN expected cycles for the same service (Cb), and the loaded cost of a single off-spec batch (B).
Per-cycle vessel cost becomes unit price divided by cycles to failure. If graphite at index 1.0 delivers 100 cycles and BN at index 4.0 delivers 500 cycles, per-cycle cost drops from 0.010 to 0.008 — BN wins on replacement alone. Add one avoided off-spec batch over the BN crucible's service life and payback strengthens further.
When a single contaminated batch exceeds the acquisition cost of a dozen BN crucibles, the 3-5× premium stops being a cost discussion and becomes an insurance calculation.
Manual cleaning time carries more weight than spreadsheets usually capture. Customers running aluminum alloy casting lines often report that cleaning labor drops before service-life savings become visible on the replacement budget — the operator-hour saving is what closes the procurement case fastest.
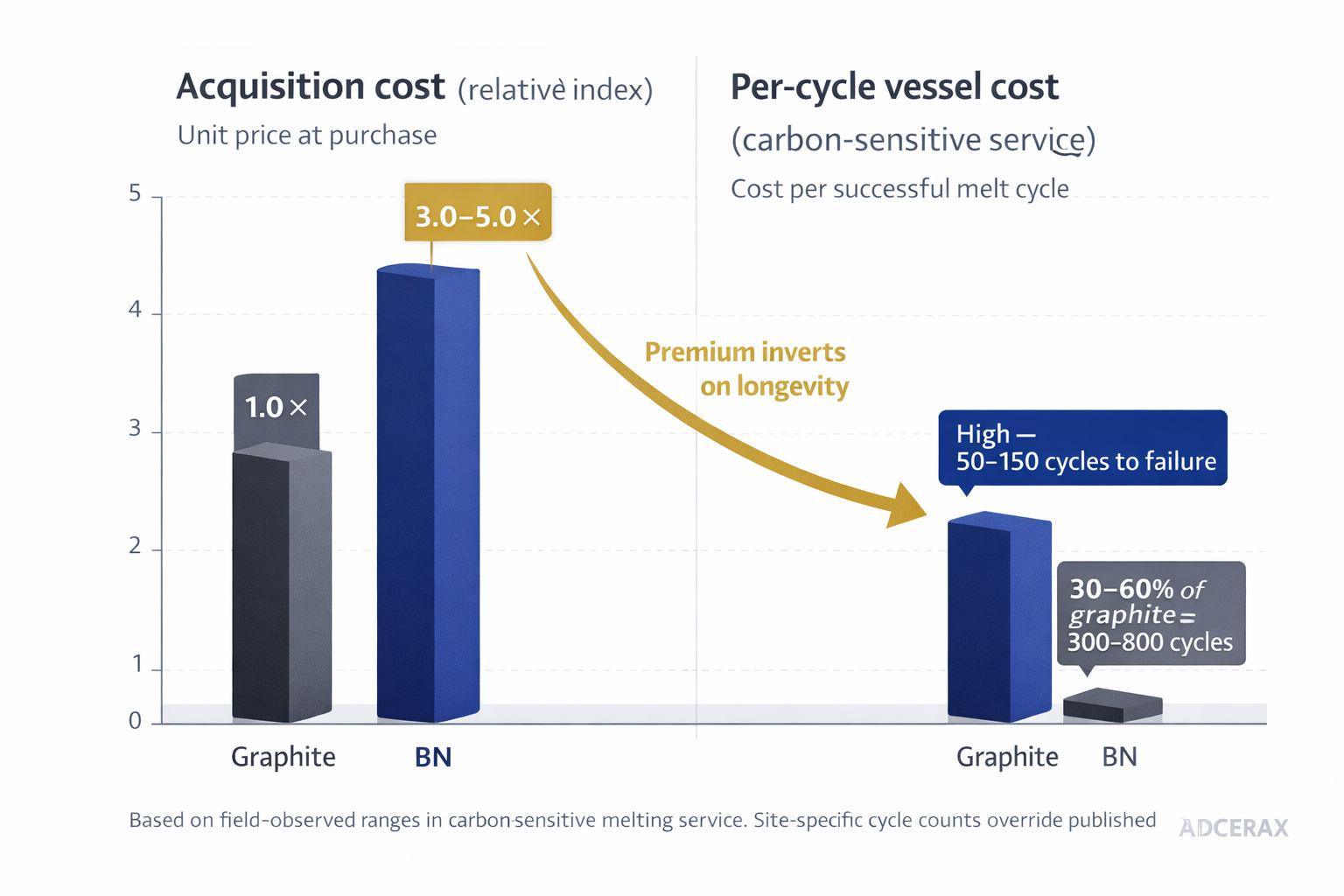
Per-cycle cost is the only figure that resolves the procurement objection — acquisition price alone tells the wrong story in carbon-sensitive service.
When BN payback never arrives — graphite-favorable cases
Three patterns eliminate payback entirely. First: inert-atmosphere melting of carbon-tolerant metals at moderate temperature. Graphite delivers full service life with no contamination loss, and BN's higher acquisition cost has nothing to amortize.
Second: short-campaign processes where the crucible is consumed within one or two charges anyway. Replacement frequency advantage vanishes.
Third: applications with low off-spec batch cost or wide quality tolerance. The contamination insurance premium loses its value.
Procurement justification template — three numbers to collect
Before proposing a switch, document Cg from the maintenance log, estimate Cb from supplier data or pilot testing, and cost a single off-spec batch including material, labor, and downstream rework. Those three numbers convert the argument from ""BN is better"" to ""BN recovers the premium in X cycles."" Procurement can validate the math; technical claims alone rarely clear the PR.
Which BN grade, density, and form factor matches your process?
Boron nitride is not one material but a family with order-of-magnitude differences in purity, cost, and performance. Hot-pressed BN at 1.9-2.1 g/cm³ density handles most molten-metal melting and inert-atmosphere crystal growth at the lower end of the price band. Pyrolytic BN, produced by CVD, delivers above 99.99% purity and near-theoretical density for MBE effusion cells and sub-100-ppm applications, at the upper end. BN composites trade some non-wetting behavior for mechanical or oxidation gains in specific industrial niches.
HBN, PBN, and BN composites — when each is the right choice
Hot-pressed BN (HBN) is the workhorse. It machines to tight tolerances, handles most carbon-sensitive melts, and represents the accessible end of the BN cost range. For aluminum melt refining, small-scale gallium evaporation, and inert-atmosphere alloy work, HBN is the default.
Pyrolytic BN (PBN) exists for a narrower purpose. Produced by CVD on a graphite mandrel, it delivers metallic impurities below 100 ppm — often below 10 ppm in premium grades — with anisotropic thermal conductivity. MBE source materials, III-V compound growth, and single-crystal growth of optoelectronic materials require PBN, not HBN.
BN composites (BN-AlN, BN-SiC, BN-ZrO₂) sit between the two. AlN additions improve thermal conductivity and mechanical strength; SiC additions improve oxidation resistance at higher temperatures; ZrO₂ additions toughen against thermal shock. Choose composites only when a documented process constraint rules out straight HBN — boron nitride material grades list the available composite systems and their target failure modes.
Density, purity, and machining tolerance — what to specify
Specify density as a minimum (g/cm³), not a target. Higher density correlates with lower open porosity, reduced melt infiltration risk, and improved mechanical strength per ASTM C1161 flexural testing.
Specify metallic impurities as a maximum ppm value — not ""high purity,"" which suppliers interpret differently. For PBN, total metallic impurities below 100 ppm is standard; premium grades reach below 10 ppm.
Form factor matters almost as much as grade. Net-shape pressed parts cost less but carry looser tolerance; machined-from-billet parts hit tight tolerance at higher price. Match the specification to geometry complexity, not to a default preference.
Handling and installation differences from graphite
BN is more brittle than graphite under impact — dropped crucibles crack rather than chip. Storage matters: unsealed HBN absorbs atmospheric moisture into binder phases, and a bake-out (~200°C for two hours before first use) prevents steam-induced spalling on the first heat.
Thermal shock tolerance is strong but not unlimited. Ramp rates above ~50°C/min on cold starts stress both HBN and PBN unnecessarily. Match installation clearances to thermal expansion — BN's coefficient is lower than graphite's, and oversized mounting compensation can create rattle during ramping.
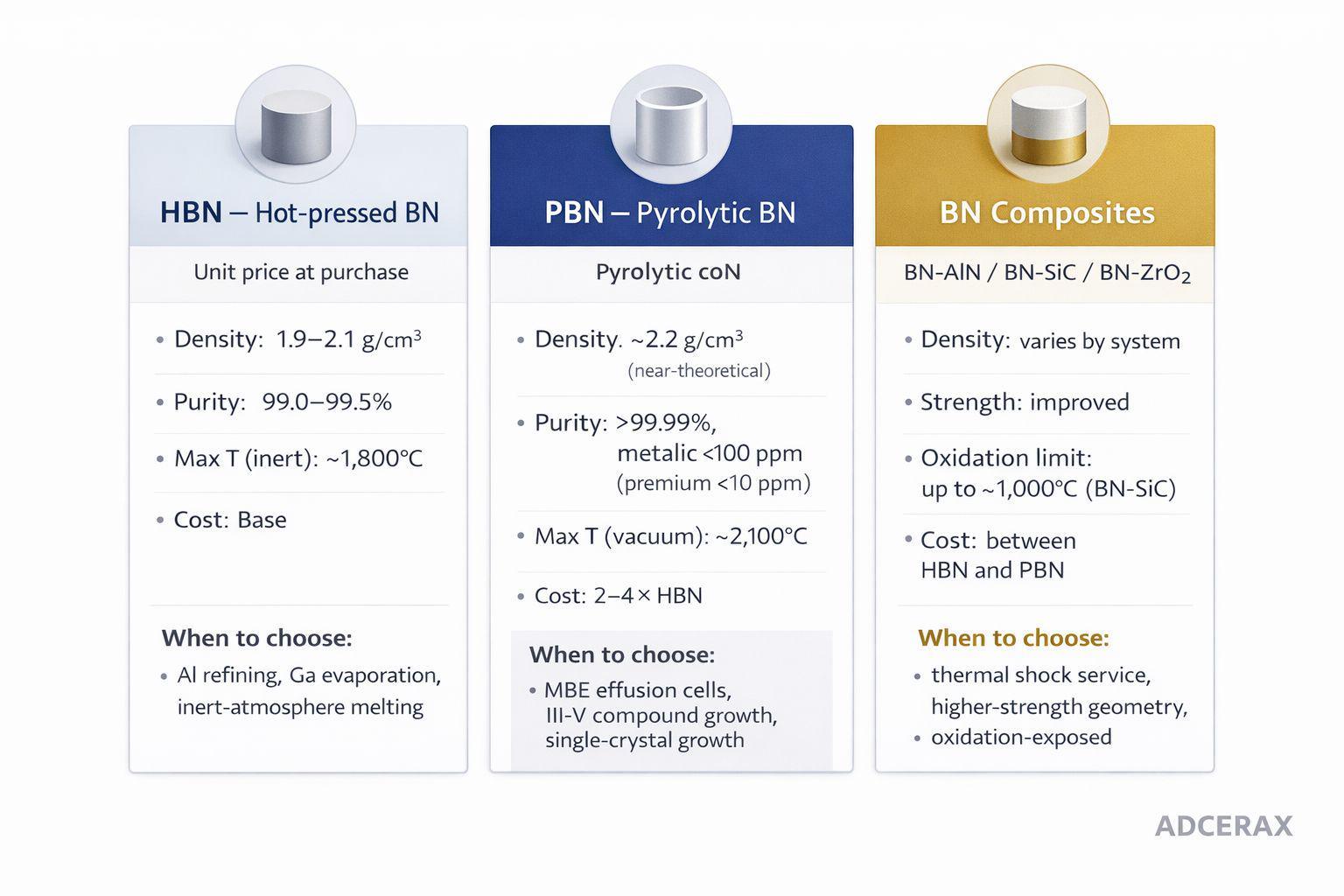
Grade selection is a three-way decision — HBN covers the bulk of carbon-sensitive melting, PBN is reserved for sub-100-ppm purity targets, composites solve specific secondary constraints.
How do you convert this decision into a vendor-ready BN crucible RFQ?
A complete RFQ needs eight fields and a four-gate sanity check. The fields: grade (HBN / PBN / composite), minimum density, maximum metallic impurities in ppm, inner geometry with tolerance, outer geometry with tolerance, surface finish, operating atmosphere, and peak temperature with cycle profile. The sanity check confirms the decision survived all four prior sections. If any gate fails, the RFQ is premature and graphite likely remains the right answer.
The eight-field RFQ template
Fill each field against the supplier's quote form:
- Grade: HBN / PBN / named composite
- Minimum density (g/cm³): specify floor, not target
- Maximum metallic impurities (ppm): total and critical elements if known
- Inner geometry with tolerance: ID, depth, taper, radius
- Outer geometry with tolerance: OD, height, flange if any
- Surface finish: as-pressed / machined Ra in µm
- Operating atmosphere: air / inert (species) / vacuum / reducing
- Peak temperature and cycle profile: hold temperature, dwell time, ramp rate, cycles per month
Three-question supplier qualification checklist
Before placing the PO: does the supplier provide a density and metallic-purity certificate per lot; have they machined this grade in this geometry previously; can they name a referenceable application close to yours? A ""no"" on any of the three is a gate, not a warning.
For related containment outside the crucible line, lab-scale ceramic vessels cover broader holding, transfer, and analysis geometries where crucible-grade BN is not required.
Conclusion
The BN-versus-graphite decision is binary on two axes and financial on a third. Atmosphere above ~500°C in air or any carbon-sensitive melt chemistry makes BN technically required regardless of cost. Below those boundaries, graphite remains the rational choice and the premium does not recover. The TCO math only runs cleanly when technical necessity is already confirmed — reversing that order generates the procurement pushback most engineers have already experienced. Specify the grade, collect the three cycle-life numbers, and the RFQ writes itself.
For engineers translating this framework into a specification, ADCERAX manufactures hot-pressed BN and PBN crucibles across the grade and geometry ranges discussed here. Share your operating atmosphere, peak temperature, melt chemistry, and target cycle life, and the technical team can return a grade recommendation with a quotation structured around the eight-field RFQ template above.
Frequently Asked Questions
What is the actual maximum operating temperature of a BN crucible in air versus inert atmosphere?
Hot-pressed boron nitride holds structural stability to approximately 850°C in air before oxidation becomes meaningful, per JIS R1648. Under inert gas cover it extends to roughly 1,800°C, and under high vacuum up to around 2,100°C. A published temperature rating always needs atmosphere context — a single number without atmosphere is incomplete.
Why are boron nitride crucibles 3 to 5 times more expensive than graphite?
The cost gap reflects synthesis route and feedstock purity. Hot-pressing requires high-purity BN powder consolidated under uniaxial pressure at roughly 1,800°C in inert gas. Pyrolytic BN adds a CVD step on a graphite mandrel, driving cost higher. Graphite, by contrast, uses lower-purity feedstock and well-established pressing lines.
What is the difference between HBN and PBN crucibles, and when does PBN become necessary?
Hot-pressed BN reaches 1.9-2.1 g/cm³ and serves most molten-metal melting in inert atmospheres. Pyrolytic BN achieves near-theoretical density and metallic impurities below 100 ppm, often below 10 ppm in premium grades. PBN becomes necessary for MBE source materials, III-V compound growth, and applications where sub-100-ppm metallic purity is non-negotiable.
What handling precautions does BN require that graphite does not?
Three differences matter operationally. Boron nitride is more brittle under impact, so drop protection during installation is essential. Unsealed HBN absorbs atmospheric moisture into binder phases, making a 200°C bake-out before first heat standard practice. Thermal ramp rates should stay below approximately 50°C per minute on cold starts to avoid unnecessary shock stress.
"





